SERVICES
Specialized technical consulting and GLP-certified preclinical studies to characterize, optimize, and validate new materials for bioprinting applications, ensuring feasibility even in the absence of pre-existing protocols
Makby Materials Lab

Makby Materials Lab provides specialized technical consulting designed to characterize, optimize, and validate new materials for bioprinting applications, specifically for scientists who need to assess the feasibility of their biomaterials even without pre-existing protocols.
Our workflow includes the development and optimization of customized printing protocols, the fabrication and testing of scaffolds under defined parameters such as geometry and environment, and the comprehensive evaluation of structural stability and post-printing performance.
Makby Materials focuses on material engineering and formulation optimization by analyzing viscosity, crosslinking, mechanical strength, and biocompatibility, delivering comprehensive technical reports with results, recommendations, and detailed operational guidelines
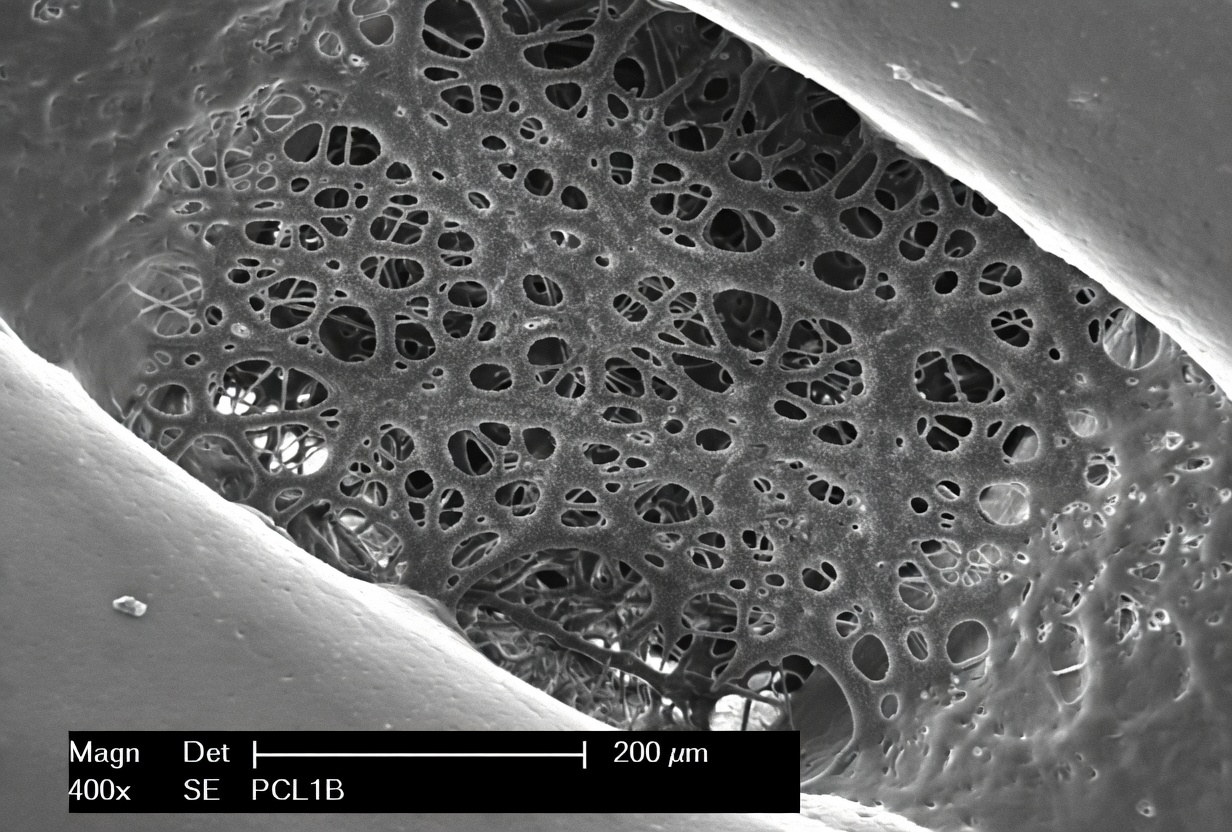
Leveraging our proprietary laser-scanning nonlinear microscopy platform, we provide high-resolution, label-free structural mapping combined with multi-channel fluorescence analysis to investigate scaffold evolution and cell-matrix interactions.
These advanced characterization services allow for the structural mapping of decellularized 3D bioprinted scaffolds to ensure design fidelity, porosity, and structural integrity, as well as the dynamic mapping of cell-laden constructs to monitor temporal evolution, collagen fiber orientation, and cell viability.
Our advanced characterization capabilities include spectroscopy via FTIR, Raman, UV-Vis, and NMR, as well as microscopy through SEM, TEM, AFM, and MEB, alongside the analysis of thermal properties using TGA, DSC, and DMA.
We evaluate mechanical properties through tensile, compression, fatigue, and rheology testing, in addition to performing surface analysis via XPS and contact angle measurements.
Makby Pre-Clinical

Robust preclinical in vivo studies are essential for validating the safety, functionality, and translational potential of technologies such as 3D bioprinted scaffolds, hydrogel patches, implantable regenerative constructs, and bioengineered medical devices.
Makby Pre-Clinical provides tailored in vitro and in vivo studies specifically designed for next-generation biomaterials and implantable medical devices to accelerate the translation of your research. Our services encompass
- Safety and biocompatibility validation to ensure safe interactions between biomaterials and host tissues.
- Functional performance assessments covering tissue integration, neovascularization, and mechanical stability.
- Biomaterial degradation kinetics and long-term implant functionality.
By bridging the gap between in vitro experimentation and human clinical application, these in vivo models provide predictive biological responses within complex living systems and generate structured datasets aligned with regulatory expectations from agencies such as the U.S. Food and Drug Administration and the European Medicines Agency.

Partnering with Makby Pre-Clinical offers access to GLP-certified expertise in toxicity, tolerance, pharmacodynamics, and biocompatibility of healthcare products, supported by ISO 9001:2015 quality standards and AAALAC-accredited animal care. We utilize cutting-edge tools including minimally invasive procedures like:
- Laparoscopy and microsurgery.
- Robotic-assisted surgery with systems such as Da Vinci XI.
- Advanced imaging technologies like 3 Tesla MRI and ultrasound.
- AI-driven multimodal data (imaging, omics) for 3D cellular models, enabling the prediction of biomaterial interactions and tissue integration.
Our tailored solutions include a wide range of clinically relevant small and large animal models, including rodents, lagomorphs, swine, sheep, and canine models, specifically designed for your technology’s needs, creating a fast-track technology translation platform that turns your concept into a clinically validated solution while optimizing scaffold architecture and controlled degradation profiles.

